In the early 1800s, there was a lot of talk among the scientific community about a new technology that facilitated the miniaturization of some elements – that is, this new technology could create “nano” materials. After years of debate, conceptualization, and development, this vague idea emerged as a highly promising field called Nanotechnology.
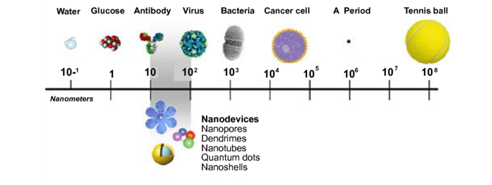
Nanotechnology is the field of science that studies nanoparticles, which are particles whose dimensions measure 100 nanometres or less. An interesting fact is that, reducing a particular element to nanosize caused dramatic changes in the basic elemental properties, thus vastly increasing its value. Over the years of its development, nanoparticles have been applied to create a massive number of new technologies. Let us have a look at how nanoparticles are synthesized.
Nanoparticles are synthesized based on two approaches – the top-down approach and the bottom-up approach. The top-down approach starts with the bulk form of the material and through various processes, the bulk matter is split into tiny parts till nanoparticles are formed. The bottom-up approach is quite the opposite, where we begin with the tiniest possible particle, the atom and nanoparticles are built by the organized assembly of these atoms.
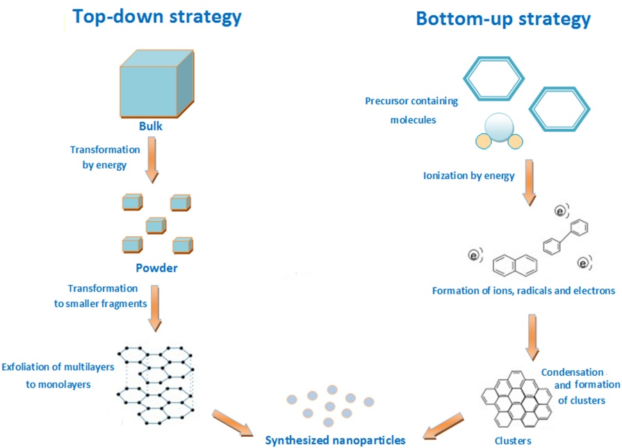
(https://iopscience.iop.org/article/10.1088/2043-6254/aa84d4)
Based on these two approaches, several methods of synthesis of nanoparticles have been devised. We can broadly classify these methods into three categories – physical methods, chemical methods, and biological methods.
As the name suggests, the Physical methods of nanoparticle synthesis involve the application of physical forces to derive nanoparticles from bulk materials. For example, the High energy ball milling and Melt Mixing methods involve the application of mechanical forces, while other methods use thermal and electrical energy, melting, evaporation and other physical forces to generate nanomaterials. Due to the nature of the process involved, most of the physical methods follow the top-down approach of synthesis. Having said that, there are a few physical methods that follow the bottoms-up strategy too. These methods mostly involve chemical reactions that are induced by physical forces such as ionization and pyrolysis. A few examples of such methods include –Inert gas condensation and pyrolysis.
The Biological methods of synthesis offer a unique advantage of being eco-friendly. These methods use the metabolic power of microorganisms, plants, and biomolecules to generate nanoparticles. Several microbes including fungi, bacteria, and algae are known to generate metal nanoparticles. Based on where the nanoparticles are synthesized, we can classify the methods into extracellular and intracellular synthesis. In the intracellular methods of synthesis, the metal ions enter the microbial cell and the aid of the cellular enzyme in the synthesis of nanomaterials. On the other hand, in extracellular synthesis, enzymes simply act on metal ions trapped on the cell surface. Another known biological method of synthesis entails the use of biomolecules, such as nucleic acids and bio-membranes, as a template to synthesize nanoparticles. For example, DNA is widely used for the synthesis of gold and silver nanoparticles. Plant-based synthesis of nanoparticles is widely acknowledged to be one of the quickest and most efficient methods. This is because the photosynthesis kinetics of nanoparticles is extremely favorable. Over the years, several plants have been discovered to have this potential. For example, nanoparticles have been successfully derived from plants like tamarind, Aloe vera, curcumin, and alfalfa, among others.
The Chemical methods of synthesis of nanoparticles happen in two distinct phases. In the first phase, the bulk metals are reduced to the precursor atoms with the help of some reducing agents. In the second phase, the atoms undergo a process called nucleation, where they are broken down, and then a slow growth process generates nanomaterials. There are three major chemical processes that have been developed – the Microemulsion process, the Sol-gel process, and Polyol process. The Microemulsion technique uses surfactants such as CTAB to stabilize microemulsions of ‘water-in-oil” or ‘oil-in-water’. The phases which are generated due to these interactions provide perfect conditions for nanoparticle synthesis. The Sol-gel process uses the chemical reactions of hydrolysis, condensation, and thermal processes to facilitate the interaction of metal precursors and the reagents to modify the shape of the nanoparticles. The Polyol method creates nanoparticles by the reduction of bulk metal precursors with the help of an alcoholic catalyst. Iron-platinum bimetallic nanoparticles are synthesized using this method.
All of these methods have aided in creating nanoparticles of high value and subsequently expanding the scope of the technology they have been applied to. Nanoparticles find extensive application in the field of medicine, where they are designed to detect tumors, modify immune responses and detoxify harmful free radicals. In the field of manufacturing, nanoparticles are used to create durable, yet lightweight materials. They have also been used to improve the biodegradability of waste.
Even today, more than 35 years into its inception, nanotechnology is still a subject of great speculation and the field holds great promises. Hopefully, within a few years’ time, science will have the ability to unlock the full potential of this technology.
References:
- https://www.slideshare.net/priyananda12/synthesis-of-nanoparticles-physicalchemical
- https://www.news-medical.net/life-sciences/Synthesis-of-Nanoparticles.aspx
- https://www.britannica.com/science/nanoparticle
- https://www.understandingnano.com/nanoparticles.html
- https://pubs.rsc.org/en/content/articlelanding/2015/ra/c5ra19388e#!divAbstract
- https://www.researchgate.net/publication/273203342_A_Review_on_Nanoparticles_Their_Synthesis_and_Types
- https://www.sciencedirect.com/science/article/pii/S1570002X0800205X