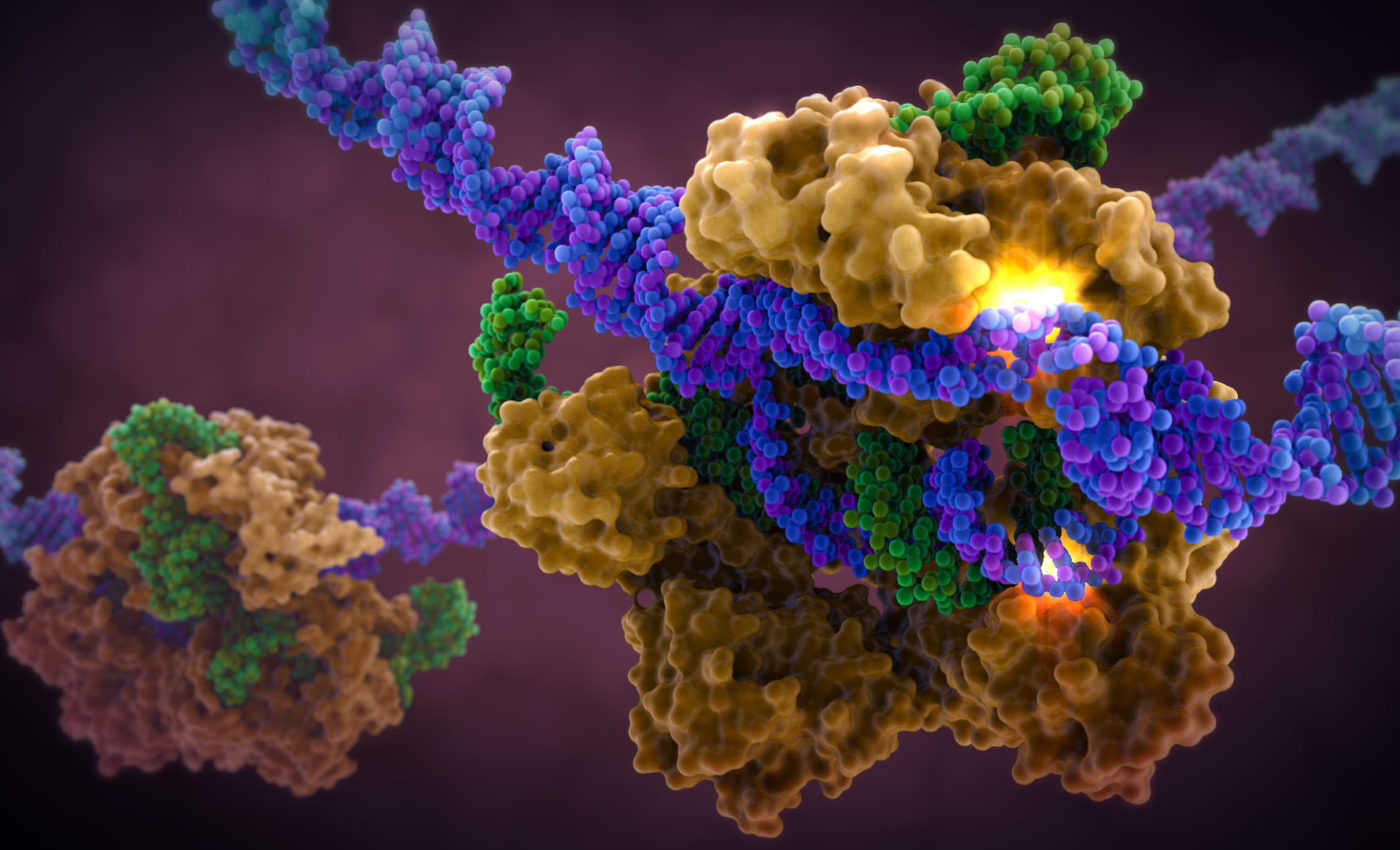
Editing the genes either its replacement or deletion is one the major working application of Biotechnology. It is very much important in designing experiments and answering questions pertaining to various scientific fields. Also in the designing of a new brand of products like the Genetically Modified Organisms (see our blog Could Mustard be India’s second genetically modified crop to be cultivated?). Various methods have been employed in gene editing but in recent times what has completely dominated the field are the CRISPR/CAS-9 systems. According to Jacob Sherkow a New York Law School patent lawyer, “Gene editing technologies come and go – this one (CRISPR/CAS-9 or CRISPR Cas 9) really seems like it has sticking power,” as told to Bloomberg. So what is it about CRISPRs that makes them so unique?
CRISPR stands for ‘Clustered Regularly Interspaced Short Palindromic Repeats’. The name justifies quite accurately what it actually is. First discovered in the bacteria Escherichia coli, it contains clusters of DNA which are identical. And in between these clusters exists DNA which is non-identical and absolutely unique. It was later found that these unique sequences are identical with the viruses, especially the viruses which attack bacteria (also known as bacteriophages). Eventually, the CRISPR-associated or ‘Cas’ genes were discovered which encodes Cas protein. These proteins are mostly helicases (unwinds the DNA) or nucleases (cuts the DNA). The sense was made from this system by considering it as a part of the bacterial immune system where a complex of Cas proteins and an RNA transcribed by the CRISPR together attach and breaks apart the invading viral DNA. A copy of the unique viral DNA may then be added to the bacterial CRISPR system. This is exactly how a human immune system works in terms of antibodies and memory B cells. This CRISPR system was however modified by two scientists, Jennifer Doudna of University of California, Berkeley and Emmanuelle Charpentier of Max Planck Institute for Infection Biology, to make it useful as a genetic tool. So how does it work?
A synthetic guide RNA (gRNA) composed of a “scaffold” sequence necessary for Cas9-binding was introduced. The user now only needs to order the spacer or targeting sequence that locks on to whatever gene they’re interested in and then mixing it up with some Cas enzyme. This will automatically cut and delete the target sequence. If another gene is to be added then a host RNA is used which will attach to the point where Cas-9 has cut the sequence. So this technology can easily be used to eliminate genes which are responsible for diseases. It can even be used to correct muscular dystrophy, target HIV virus or produce the designer baby (genetically modified human embryo).
The working of CRISPR-Cas9
(Source: origene.com)
There are not all goods to the CRISPR story. It can very well lead to off-target mutations (secondary mutations in regions not targeted by the guide RNA (gRNA)). In fact, a very recent article published in the Newsweek mentions research done on mice wherein the use of ‘CRISPR technology had accomplished the originally intended task of correcting a gene that causes blindness, however, it had also resulted in 1,500 other small changes and 100 large changes. Not a single of those changes had been predicted by the researchers’. Not to mention the vast range of ethical concerns when embryos could be mutated according to the desire of the man.
Jennifer Doudna (above) and Emmanuelle Charpentier (below)
(Source: (above)carnegiescience.edu and source (below)medfak.umu.se)
The co-inventor of CRISPR Jennifer Doudna in a TED talk given in the year 2015 said that she along with her colleagues wants to have a global conversation on the issues arising from the use of CRISPR-Cas9. She asserted that given all its potentials and possible risks a prudent path towards the employment of this technology is necessary.
References:
- www.newsweek.com/unexpected-mutations-new-gene-editing-technology-crispr-618235
- www.youtube.com/watch?v=TdBAHexVYzc