Age-related macular degeneration (AMD) or simply macular degeneration is a phenomenon characterized by loss of vision or blurriness in the centre of the visual field. Some advanced forms of AMD are referred to as Geographic Atrophy. Although it is different from complete blindness it can make everyday activities like recognizing faces, driving or handling devices very difficult. The degeneration or damage occurs in the macula of the retina which is an oval-shaped pigmented area near the centre of the retina. The pathophysiology of AMD is not yet established and theories often suggest oxidative stress, mitochondrial dysfunction, or genetic studies implicating the dysregulation of the alternative complement pathway. Likewise, the exact causes of the condition are also unclear. Some common risk factors are age, smoking, and genetic makeup. Nonetheless, this condition affects over 6.2 million people globally and is the leading cause of vision loss amongst the elderly. Genentech, Inc., a biotechnology company based in the United States tested her drug lampalizumab on AMD patients. The drug slowed down the destruction of photo-receptors or light-sensing cells in the retina.

Lampalizumab is a synthetic antigen-binding fragment of a humanized monoclonal antibody. The study comprised of administering it as eye injections into about 129 patients for a period of about 18 months. After one month there was 20% reduction in the lesion area progression and 44% reduction in geographic atrophy damage as measured by fundus autofluorescence in the patients which took the drug as compared to the untreated controls. However, it is crucial to note that these changes were not observed in every candidate receiving the drug but only in 43%.
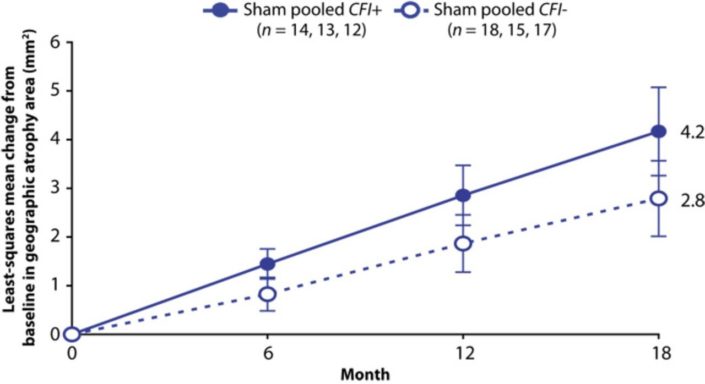
(Source: Yaspan et al. 2017)
This 43 % of the tested population was actually the patients in which the complement factor I (CFI) risk allele carrier was absent. CFI polymorphisms can alter CFI production which has been in the past well established for being directly associated with a high risk of age-related macular degeneration. It is in these patients (CFI- ) that Lampalizumab targets complement factor D, an important activator of the alternative complement pathway which helps in fighting the infection.
This study conducted by Genentech research team was reported in the journal Science Translational Medicine. No significant side effects were observed. While the study is too small to prove if lampalizumab really helps maintain vision in AMD it provides a guide to design much larger studies which can further test the therapeutic potentials of the drug.
Reference:
Brian L et al, 2017. Targeting factor D of the alternative complement pathway reduces geographic atrophy progression secondary to age-related macular degeneration. Science Translational Medicine.